 |
 |
- Search
| Ann Pediatr Endocrinol Metab > Volume 19(4); 2014 > Article |
Abstract
Purpose
The diagnosis of pituitary stalk lesion has been based on clinical feature, radiologic assessment for its critical location and role. This study aimed to investigate clinical symptoms, endocrine disturbance, magnetic resonance imaging (MRI) findings of pituitary stalk lesions in children and adolescents and to evaluate differences between neoplastic lesions with the others.
Methods
We performed a retrospective review of patients under 18 years old with pituitary stalk lesions diagnosed at the Seoul National University Children's Hospital between 2000 and 2013, by a text search for head MRI reports by using 'pituitary stalk', 'infundibulum', and 'infundibular stalk', as keywords.
Results
For the 76 patients, sixteen patients (21.1%) had congenital lesions, and 52 (68.4%) had neoplasms. No inflammatory lesions were found. Diabetes insipidus (DI) was the most common endocrine defect, diagnosed in 38 patients (50%). There was male predominance especially in neoplastic group. Thickened pituitary stalk was, but enhancement of lesion was not, associated with neoplasm. DI was more prevalent in neoplastic stalk lesions. Anterior pituitary dysfunction such as growth hormone and adrenocorticotropic hormone deficiencies were less prevalent in neoplastic lesions of pituitary stalk.
The pituitary stalk, known as the infundibulum or infundibular stalk, is the connection between the hypothalamus and the pituitary gland, especially the posterior pituitary gland. The shape of the stalk is smoothly tapering, and the normal pituitary stalk is widest superiorly, measuring 3.25±0.56 mm in transverse diameter at the optic chiasm and 1.91±0.40 mm at its pituitary insertion1). Signal intensity of the pituitary stalk is less than that of the optic chiasm and posterior pituitary gland on brain magnetic resonance imaging (MRI) T1-weighted images. Deviation/tilt of the pituitary stalk can be seen without any problems1). Disturbance of the connection between the hypothalamus and the pituitary gland via pituitary stalk can cause anterior and posterior pituitary dysfunction. Because of the critical location and role of the pituitary stalk, patients who have disease involving the pituitary stalk often present with various clinical features of hypopituitarism, DI, or hyperprolactinemia, and clinicians have hesitated to perform biopsies that may cause iatrogenic hypopituitarism. Therefore, the diagnosis of pituitary stalk lesions has been based on not only pathologic examination but also clinical and radiologic evaluations, such as MRI.
Pituitary stalk lesions can be divided into the following 3 categories: congenital/developmental, inflammatory/infectious, and neoplastic2). It is important for clinicians to discriminate neoplastic lesion from the other lesion in terms with treatment and prognosis.
A few studies of pituitary stalk lesions have been performed; however, most of the study subjects were adults with only a small number of children3,4). Herein, we reviewed the clinical features, radiologic findings, and pathologic diagnoses of pituitary stalk lesions in children and adolescents treated at the Seoul National University Children's Hospital. Also, we investigated the difference of clinical and radiologic findings between the neoplastic and nonneoplastic lesions of pituitary stalk.
This study is a retrospective review of patients under 18 years of age with pituitary stalk lesions, diagnosed and treated at the Seoul National University Children's Hospital between January 1, 2000, and June 30, 2013. We performed a text search for head MRI reports by using 'pituitary stalk', 'infundibulum', and 'infundibular stalk', as keywords, and a total of 231 patients were found. Of these, 152 patients with a normal pituitary stalk, described as 'normal pituitary stalk' or 'normal infundibulum' in their MRI report, were excluded. In addition, 1 patient who had an MRI report only without clinical findings was excluded and 2 patients whose MRI abnormality was postoperative change were excluded. In the remaining 76 patients, we characterized the pituitary stalk lesions by using the initial brain MRI on which a stalk abnormality was identified (Fig. 1). All laboratory, pathological, and clinical evaluation, were reviewed. We compared the clinical, radiological, and laboratory findings of neoplastic group and nonneoplastic group.
A pathologic diagnosis was obtained using tissue from (1) the pituitary stalk itself, (2) other areas of the brain in which the MRI findings were analogous to the stalk's, or (3) an extracranial site when clinical evaluation, laboratory, and radiologic findings highly suggested pituitary stalk involvement of a specific disease, such as metastatic tumor or inflammatory disease. Tissue was obtained via biopsy or excision of the lesion of interest. When a specific disease was strongly suspected, a diagnosis was made without pathological analysis.
We divided the whole subjects into two groups by the etiology of pituitary stalk lesion; neoplasm and nonneoplasm. Patients with congenital anomalies, inflammatory/infectious lesions, and undiagnosed lesions were included in nonneoplastic group.
Low levels of the primary hormones along with low or abnormal pituitary trophic hormones implied secondary hormonal deficiencies. The adrenocorticotropic hormone (ACTH) stimulation test was often used for confirmation of adrenal insufficiency. In some patients, growth hormone (GH) and insulin-like growth factor 1 (IGF-1) were simply measured without further confirmatory tests. DI was diagnosed based on typical clinical features or the measurement of serum, urinary osmolality, and electrolytes. Water deprivation tests were occasionally performed. Hormonal status was evaluated before and after biopsy, excision, or local treatment such as radiation of the lesion.
IBM SPSS Statistics ver. 21.0 (IBM Co., Armonk, NY, USA) was used for statistical analyses. A value of P≤0.05 was considered statistically significant. We used the paired t-test for comparing means of continuous variables. The Pearson chi-square test and Fisher exact test were also used to compare proportions between groups.
Pituitary stalk lesions were classified according to etiology as congenital in 16 patients (21.1%) and neoplastic, including Langerhans cell histiocytosis (LCH), in 52 patients (68.4%). No inflammatory or infectious lesions were seen. In 8 patients (10.5%), the cause of the pituitary stalk lesion remained unknown. Of the congenital anomalies, ectopic posterior pituitary gland was the most common, accounting for 50%. Of the neoplastic lesions, the most common etiology was intracranial germ cell tumor, accounting for 40.4%, and the second most common was LCH, accounting for 28.8%. Intracranial germ cell tumors and LCH accounted for 69.2% of neoplastic lesions and 47.3% of all pituitary stalk abnormalities in our cohort (Table 1).
Diagnosis was confirmed via pathological evaluation with biopsy or excision in 37 patients (48.7%). Tissues were obtained from the pituitary stalk or other areas of the brain in which the MRI appearance was similar to the stalk or extracranial site of interest. Of the 37 patients with pathological evaluation, 13 patients had intracranial germ cell tumor, 8 patients had LCH, 4 patients had medulloblastoma. 1 had unknown etiology and the others had various neoplasms described in Table 1.
Of the 76 patients, 49 (64.5%) were male and 27 (35.5%) were female patients. The mean age at diagnosis was 8.80±4.83 years (1 month-18 years). The neoplastic lesion was more prevalent in boys than in girls (Table 2). The most common presenting symptoms was polyuria and/or polydipsia (n=33, 43.4%), followed by short stature (n=12, 15.8%), visual disturbance (n=11, 14.5%), seizure and/or altered mentality (n=10, 13.2%), headache (n=8, 10.5%), abnormal puberty (n=7, 9.2%), vomiting (n=4, 5.3%), hypothyroidism (n=4, 5.3%), gait disturbance (n=2, 2.6%), hypoglycemia (n=4, 5.3%), and hearing loss (n=1, 1.3%).
Compared with nonneoplastic group, neoplastic patients showed more prevalent symptoms of polyuria/polydipsia, and less prevalent short stature and hypoglycemia (Table 2).
The MRI findings of pituitary stalk lesions were change of stalk thickness, no visualization, and abnormal high signal intensity. Increased thickness was the most common finding, occurring in 69.7% (53 patients, including 36 with unenhanced/unchanged signal intensity of stalk). In 9 patients (11.8%), the pituitary stalk was not visualized, and in 3 patients (3.9%), the stalk thickness decreased. Eleven patients (14.5%) showed only an enhanced pituitary stalk without change in thickness. Of the 53 increased lesions, 48 (90.6%) were neoplastic; of the 12 decreased or not visualized lesions, 9 (75%) were congenital, and the remainder were of unknown etiology. Increased stalk thickness was associated with neoplastic lesions significantly (P<0.001) (Table 3).
Twenty-nine patients (38.2%) had pituitary stalk lesion with high signal intensity and 47 patients (61.8%) without enhancement. There was no significant association between neoplastic lesion and pituitary stalk enhancement (Table 3).
The extent of pituitary stalk lesions ranged from confinement to the stalk to extension to an extracranial site. In 7 patients (9.2%), the lesions were limited to the pituitary stalk; in 30 patients (39.5%), lesions were within the pituitary stalk and sellar area; in 31 patients (40.8 %), lesions extended to the suprasellar or other intracranial area; and in 8 patients (10.5%), lesions extended to an extracranial site. There was also no significant association between neoplasm and limitation of lesion within pituitary stalk (Table 3).
DI was the most common endocrine abnormality, occurring in 38 patients (50% of all patients). The next most common endocrine abnormality was secondary hypothyroidism, occurring in 30 patients (39.5% of all patients, 45.5% of tested subjects), followed by low level of IGF-1 or GH and GH deficiency confirmed by stimulation testing in 23 patients (30.3% of all patients, 39.7% of tested subjects). Deficiency of the adrenal axis was diagnosed in 20 patients (26.3% of all patients, 34.5% of tested subjects), and secondary hypogonadism was diagnosed in 11 patients (14.5% of all patients, 23.4% of tested subjects). In 2 of the patients who were evaluated for gonadal axis abnormalities, precocious puberty was suggested. A total of 38 patients (50% of all patients) showed more than one anterior pituitary hormone deficiency.
GH and ACTH deficiency were less prevalent in neoplastic group than in nonneoplastic group. But central DI was more prevalent in neoplasm group. There was no significant difference in prevalence of thyrotropin and gonadotropin deficiency between neoplastic and nonneoplastic group (Table 3).
In this study, we reviewed the etiology of pituitary stalk lesions in children and adolescents evaluated at the Seoul National University Children's Hospital. Previous studies of pituitary stalk lesions have largely focused on adults or a specific disease/clinical feature, such as LCH5) or DI6), or have contained few cases.
Turcu et al.3) and Hamilton et al.4) published large case series with a focus on pituitary stalk lesions. The study of Hamilton et al.4) included 44 adults and 21 children and adolescents, and that of Turcu et al.3) included 135 adults and 17 children and adolescents. In accordance with our study, neoplastic lesions predominated in these studies regardless of the classification of LCH as a tumorous condition by Hamilton et al.4) and as an inflammatory disorder by Turcu et al.3). Therefore, when a pituitary stalk lesion is discovered and believed to be a primary malignancy, it may be necessary to evaluate stalk tissue. However, because of the risks of hypopituitarism caused by a stalk biopsy, few patients receive a pathologic diagnosis based on tissue from the pituitary stalk. In the studies of Hamilton et al.4) and Turcu et al.3), 26 of 65 patients (40%) and 37 of 152 patients (24%), respectively, had a pathologic diagnosis with tissue samples from the pituitary stalk or other sites. In our study, 37 of 76 patients (48.7%) underwent biopsy or excision at the stalk or other site. In the study of Beni-Adani et al.7), among 7 children with DI who had surgery for the pathologic diagnosis of a thickened pituitary stalk, none deteriorated neurologically or endocrinologically after the operation. The authors argued that, therefore, open biopsy should be the preferred diagnostic method in children with a thickened pituitary stalk.
In terms of endocrine disturbance in our study, half of the patients developed DI and a half had one or more adenohypophyseal hormone deficiencies. Secondary hypothyroidism was the most common, and secondary hypogonadism was the least common adenohypophyseal hormone deficiency. In the study of Turcu et al.3), secondary hypogonadism was the most common, and secondary adrenal insufficiency was the least common pituitary hormonal deficiency. The hormonal test most frequently performed was for evaluation of thyroid function, and the least performed test was for evaluation of GH.
Congenital lesions were found most frequently in children in the series of Hamilton et al.4). In the study of Turcu et al.3), no specific category predominated in pediatric patients. In our study, neoplasm, including LCH, was the most common etiology of pituitary stalk lesions, accounting for 68.4%. Next, congenital lesions accounted for 21.1%. However, because Seoul National University Children's Hospital is a tertiary hospital where many children are treated for neoplasm, selection bias may have occurred. We classified LCH as a neoplasm because it was sometimes difficult to distinguish on MRI between an intracranial tumor, such as an intracranial germ cell tumor, and LCH. Three patients were diagnosed with acute lymphoblastic leukemia/lymphoma (1 patient with acute lymphoblastic leukemia, L1; 1 patient with precursor B-cell acute lymphoblastic leukemia; and 1 with diffuse large B-cell lymphoma). The patient with acute lymphoblastic leukemia, subtype L1, had normal pituitary hormone status. In the patient with precursor B-cell acute lymphoblastic leukemia, pituitary hormones were not evaluated except for those of the hypothalamo-neurohypophyseal tract and in the patient with diffuse large B-cell lymphoma, tests for GH gonadal axis were not done.
In our study, we found only 1 case of metastatic solid cancer, which was a retinoblastoma with brain metastasis. In the study of Turcu et al.3), neurosarcoidosis was diagnosed in 11 patients and was the most common etiology of inflammatory lesions. In addition, in a previous study, stalk thickening and hypophyseal and hypothalamic infiltration were reported in patients with neurosarcoidosis8). Nevertheless, we did not find any cases of neurosarcoidosis. Sarcoidosis manifests most commonly in the fourth decade of life9,10,11). It occurs throughout the world in all races, and the highest annual incidence of 5 to 40 cases per 100,000 people has been observed in northern European countries12,13). Childhood-onset sarcoidosis is rare and can occur as a part of systemic sarcoidosis or as an isolated presentation9,14,15,16).
In the study of Turcu et al.3), congenital lesions had a specific pattern of enhancement on MRI, but neither the pattern of MRI enhancement nor the thickness of the pituitary stalk lesion correlated with clinical features.
There are some limitations in our study. First, it was a retrospective review. As a result, uniform testing of pituitary hormonal status was not done. Second crucial limitation is the lack of diagnoses confirmed via tissue biopsy. Because biopsy from the stalk can cause deficiencies of pituitary hormones, diagnoses were based on clinical features and imaging. The unknown pituitary lesions could be diagnosed as a neoplasm later. Third, the number of patient was too small to apply the conclusions to general population in spite of relatively large pediatric cohort comparing with previous studies. In our study, the predominant neoplastic patient number was due to selection bias as mentioned above.
Despite all these limitations, our study showed that the etiology of pituitary stalk lesions in children and adolescents was variable and slightly different from that in adults. Neoplastic lesion was common and infectious/inflammatory lesion was rare. Male predominance was found especially in neoplastic group. On MRI, pituitary stalk thickness was related with neoplasm but enhancement of lesion was not. The most common presenting symptom and hormone abnormality of pituitary stalk lesion was DI, which was more prevalent in neoplastic stalk lesions. GH and ACTH deficiencies were more associated with nonneoplastic lesions.
In conclusion, the etiology of pituitary stalk lesions in children and adolescents was variable and slightly different from that in adults. A systemic evaluation of clinical symptoms, endocrine disturbances, and radiologic findings of pituitary stalk lesion, could help to suspect neoplastic lesions which need a pathologic evaluation such as biopsy or excision.
References
1. Simmons GE, Suchnicki JE, Rak KM, Damiano TR. MR imaging of the pituitary stalk: size, shape, and enhancement pattern. AJR Am J Roentgenol 1992;159:375–377. PMID: 1632360.


2. Rupp D, Molitch M. Pituitary stalk lesions. Curr Opin Endocrinol Diabetes Obes 2008;15:339–345. PMID: 18594274.


3. Turcu AF, Erickson BJ, Lin E, Guadalix S, Schwartz K, Scheithauer BW, et al. Pituitary stalk lesions: the Mayo Clinic experience. J Clin Endocrinol Metab 2013;98:1812–1818. PMID: 23533231.



4. Hamilton BE, Salzman KL, Osborn AG. Anatomic and pathologic spectrum of pituitary infundibulum lesions. AJR Am J Roentgenol 2007;188:W223–W232. PMID: 17312027.


5. Marchand I, Barkaoui MA, Garel C, Polak M, Donadieu J. Writing Committee. Central diabetes insipidus as the inaugural manifestation of Langerhans cell histiocytosis: natural history and medical evaluation of 26 children and adolescents. J Clin Endocrinol Metab 2011;96:E1352–E1360. PMID: 21752883.


6. Di Iorgi N, Allegri AE, Napoli F, Calcagno A, Calandra E, Fratangeli N, et al. Central diabetes insipidus in children and young adults: etiological diagnosis and long-term outcome of idiopathic cases. J Clin Endocrinol Metab 2014;99:1264–1272. PMID: 24276447.



7. Beni-Adani L, Sainte-Rose C, Zerah M, Brunelle F, Constantini S, Renier D, et al. Surgical implications of the thickened pituitary stalk accompanied by central diabetes insipidus. J Neurosurg 2005;103(2 Suppl):142–147. PMID: 16370280.


8. Bihan H, Christozova V, Dumas JL, Jomaa R, Valeyre D, Tazi A, et al. Sarcoidosis: clinical, hormonal, and magnetic resonance imaging (MRI) manifestations of hypothalamicpituitary disease in 9 patients and review of the literature. Medicine (Baltimore) 2007;86:259–268. PMID: 17873755.


9. Twilt M, Benseler SM. Childhood inflammator y brain diseases: pathogenesis, diagnosis and therapy. Rheumatology (Oxford) 2014;53:1359–1368. PMID: 24324213.



10. Nozaki K, Scott TF, Sohn M, Judson MA. Isolated neurosarcoidosis: case series in 2 sarcoidosis centers. Neurologist 2012;18:373–377. PMID: 23114669.


11. Oksanen V. Neurosarcoidosis: clinical presentations and course in 50 patients. Acta Neurol Scand 1986;73:283–290. PMID: 3716768.


12. Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med 2007;357:2153–2165. PMID: 18032765.


13. Pietinalho A, Hiraga Y, Hosoda Y, Lofroos AB, Yamaguchi M, Selroos O. The frequency of sarcoidosis in Finland and Hokkaido, Japan. A comparative epidemiological study. Sarcoidosis 1995;12:61–67. PMID: 7617979.

14. Scholten V, ten Hove WM, Macdonald EA. An unusual presentation of neurosarcoidosis in an 11-year-old boy. Can J Neurol Sci 2009;36:783–786. PMID: 19960762.


15. Tsao CY, Lo WD, Rusin JA, Henwood MJ, Boue DR. Isolated neurosarcoidosis presenting as headache and multiple brain and spinal cord lesions mimicking central nervous system metastases. Brain Dev 2007;29:514–518. PMID: 17307323.


16. Baumann RJ, Robertson WC Jr. Neurosarcoid presents differently in children than in adults. Pediatrics 2003;112(6 Pt 1):e480–e486. PMID: 14654650.


Fig. 1
A total 231 patients was found whose magnetic resonance imaging (MRI) reports included key words such as 'pituitary stalk', 'infundibulum', and 'infundibular stalk'. One hundred and fifty two patients with a normal pituitary stalk in their MRI report, 1 patient who had an MRI report only, and 2 patients with postoperative change, were excluded. Of the remaining 76 patients, 16 patients had congenital lesions, 52 patients neoplasm, and 8 patients unknown etiologies.
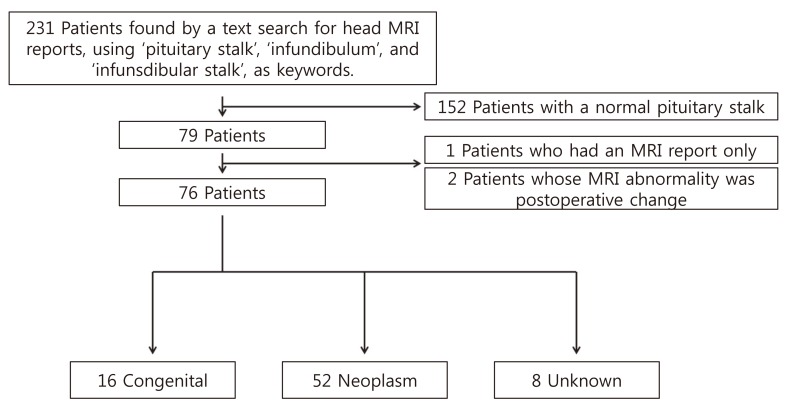
Table 1.
Etiology of pituitary stalk lesion
Table 2.
Demographic data and clinical features of pituitary stalk lesion
Table 3.
Magnetic resonance imaging findings and endocrine disturbances of pituitary stalk lesion
- TOOLS
-
METRICS

-
- 10 Crossref
- 10,037 View
- 167 Download
- Related articles in APEM
-
Hypoparathyroidism in children and adolescents2023 September;28(3)





